DSS-Induced Colitis in C57BL/6 Mice
DISCONTINUED MODEL
Effective April 2022, Hooke no longer runs or recommends this model. We did run DSS-induced colitis regularly for a number of years, but our experience was that FDA-approved colitis treatments (efficacious in human patients) are not efficacious in this mouse model, and despite many attempts we have been unable to reproduce published results in this model.
We suspect published reports of compound efficacy in this model may reflect only reduced consumption of DSS by mice, resulting in weaker colitis.
Instead, Hooke recommends CD4+CD45RBhigh-induced colitis in SCID mice.
We have left this page active for reference purposes only.
Inflammatory bowel disease (IBD)
Inflammatory bowel disease (IBD) is an inflammation of all or part of the human gastrointestinal (GI) tract. The most common forms of IBD are ulcerative colitis, which affects the colon, and Crohn's disease, which can affect any part of the GI tract (but most often colon and terminal ileum).
DSS-induced colitis
Dextran sodium sulfate (DSS) induced colitis in C57BL/6 mice shares many clinical and histopathological characteristics with human IBD. Like human patients with IBD, mice administered DSS lose weight, develop bloody diarrhea and have histopathological findings of colitis.
We induce colitis in C57BL/6 mice by administering 3 to 4 % DSS in drinking water for 5 days. The DSS causes inflammation in the entire gut, but the damage is most prominent in the colon. It has been proposed that negatively charged sulfate residues of DSS chelate divalent cations (e.g. calcium and magnesium), which are required for maintenance of epithelial cell tight junctions. Disruption of these tight junctions leads to a loss of epithelial integrity, mucosal injury and increased exposure to luminal antigens, all resulting in inflammation primarily due to activation of the innate immune response.
When DSS is removed from the drinking water, acute colitis continues to evolve into chronic inflammation [2].
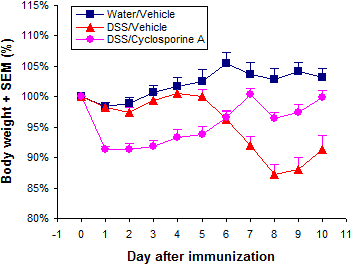
Clinically, mice develop diarrhea on Day 1 or 2 and weight loss starts on Days 5 to 7, and reaches a maximum between Days 9 and 11. Stool consistency worsens for approximately 9 days, followed by partial recovery over the next several weeks.
Blood is present in stool during the acute phase of disease, with a peak around Day 5, and is usually not found after Day 7.
Histopathologically, colitis is characterized by loss of crypts, goblet cell reduction, presence of inflammatory infiltrates, edema, and signs of epithelial regeneration and reparation.
DSS-induced colitis develops even in the absence of an adaptive immune system (i.e. in SCID or RAG-1KO mice, which lack T and B cells), indicating that inflammation in DSS-induced colitis is mediated primarily by the innate immune response. Therefore, this model of IBD is considered most useful for evaluation of compounds expected to play a role in modifying the innate immune response and in promoting epithelial proliferation, restitution and mucosal wound repair in the presence of chronic inflammation.
Please contact Hooke at or with questions or for a quotation.
Readouts
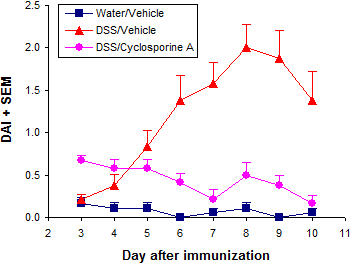
Our standard in vivo readouts are body weight and stool score, both taken daily. A daily disease activity index (DAI) is then calculated from body weight and stool scores.
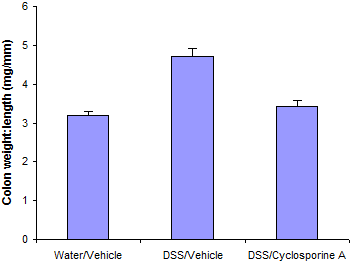
At the end of the study mice are euthanized and colons are collected, cleaned, and measured for weight and length.
Histological analysis is the main readout in these studies - colons are made into "Swiss rolls" or sectioned, fixed in buffered formalin, then histologically analyzed. Scoring is performed blind.
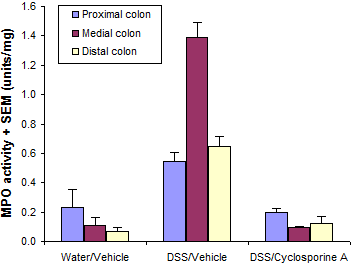
Colons are also often analyzed for MPO activity, or lamina propria cells are isolated and analyzed by flow cytometry (see below).
Typical results from a study at Hooke are shown below.
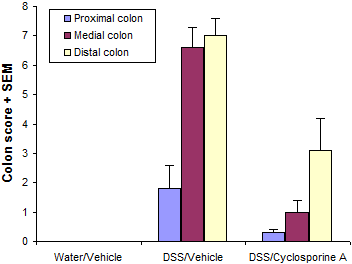
Typical histological findings in colons
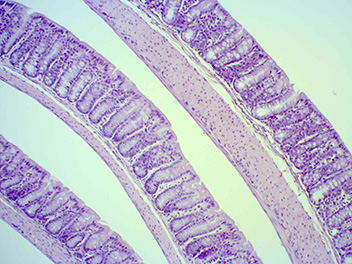
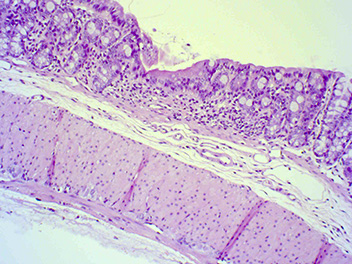
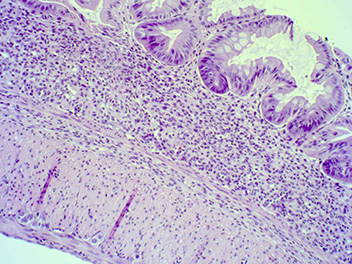
The images below illustrate typical changes in colons of mice with DSS-induced colitis.
Normal colon; DSS not administered
DSS administered, treated with Cyclosporin A
DSS administered, treated with vehicle
Flow cytometric analysis
Some customers order analysis of all groups in advance, but we generally encourage customers to wait for initial results before deciding which analyses to order.
We usually quote a price for your base study, together with prices for additional analysis that may be ordered later. This gives you flexibility to analyze only a sample of animals, choose particular groups for analysis, or even skip analysis completely if the results aren't promising. Our scientists will review your initial results with you, and can recommend the best analyses to maximize return from your study.
We generally ask for 5 day's notice to schedule tissue collection or analysis (but we do our best to accommodate last-minute orders).
Flow cytometric analysis of cells
Cells are stained and analyzed by flow cytometry. The customer selects up to 4 antibodies for each analysis. Multiple analyses can be performed on each animal.
We normally have a wide range of antibodies in stock and routinely perform 15 color analysis. We perform both surface and intracellular staining.
References
[1] Mähler M et al, Am J Physiol 274:G544 (1998)[2] Melgar S et al, Am J Physiol Gastrointest Liver Physiol. 288:G1328 (2005)
[3] Melgar S et al, Int Immunopharmacol. 8:836 (2008)


_150px.jpg)